Introduction
Stenocereus pruinosus (Otto) Buxbaum is a cactus species of columnar habit that develops in arid and semi-arid regions like those located in the Balsas river basin and Tehuacán Valley in Mexico (Luna-Morales, 2006; Martínez-Villegas et al., 2011; Parra, Pérez-Nasser, Lira, Pérez-Salicrup, & Casas, 2008). Fruit are oval berries, with epicarp that possesses deciduous thorns (Figure 1), and flesh with multiple seeds and with different colors (Luna-Morales, 2007). Consumption of this type of fruit provides nutrients like vitamins B, C, and E (Bravo-Hollis & Sánchez-Mejorada, 1991), minerals such as iron, copper, and zinc, as well as antioxidant compounds like soluble phenols and betalains (García-Cruz, Valle-Guadarrama, Salinas-Moreno, & Joaquín-Cruz, 2013). Due to the presence of some of these compounds in flesh, pitaya fruit have been classified as functional foods (Beltrán-Orozco, Oliva-Coba, Gallardo-Velázquez, & Osorio-Revilla, 2009).

Pitaya fruit have good acceptance and high potential in the fresh fruit market, but their shelf life is less than six days at ambient temperature (García-Cruz, Valle-Guadarrama, Salinas-Moreno, & Luna-Morales, 2016), and this limits their commercialization and consumption to local production areas. Additionally, the presence of thorns makes their postharvest handling difficult and such a feature can be a factor leading to mechanical damage. However, the impact of removing those anatomical structures on the quality attributes of fruit has not been assessed.
Refrigeration is considered an efficient strategy to reduce the effect of deterioration factors in horticultural products and to extend shelf life (Nunes & Emond, 2003), but so far this alternative has not been evaluated for handling pitaya fruit in postharvest. In this regard, this work was performed to evaluate the postharvest behavior of pitaya fruit under refrigeration conditions and to determine the effect of removing thorns on fruit quality attributes. Fruit of cactaceae species are sensitive to chilling injury and the optimal storage temperature has been located between 8 and 12 °C (Esquivel, 2004; Rodriguez, Casóliba, Questa, & Felker, 2005). In the present work, in order to avoid incidence of chilling injury in fruit, a temperature of 12 °C was selected to conduct the experimental storage.
Materials and methods
Plant material
Two hundred pitaya (Stenocereus pruinosus (Otto) Buxbaum) fruit with red flesh and 200 fruit with orange flesh were collected in Tepexi de Rodríguez, Puebla, Mexico (18° 35’ 46’’ NL, 97° 55’ 48 WL; 1644 masl) at commercial maturity, which occurred when thorns were easily released and skin had become bright (Figure 1).
Experimental organization
Fruit were immersed in NaClO solution (500 mg∙L-1) for 5 min in order to reduce microbial load. Thereafter, they were dried with absorbent paper and weighed. One hundred and fifty fruit that were free of defects and with uniform size were chosen from each variant. Thorns were removed from half of the fruit and the following treatments were formed: fruit of red flesh with thorns (RwT) and without thorns (RoT), and fruit of orange flesh with thorns (OwT) and without thorns (OoT). All fruit were stored at 12 °C for 21 days, during which time the relative humidity reached 76 %.
The day when storage began (0 d) five fruit were sampled from each treatment in order to make an initial characterization. Thereafter, every other day six fruit from each treatment were removed from the storage room and three experimental units with two fruit each were formed, with the purpose of carrying out a characterization in terms of weight loss, color, respiration rate, firmness, total soluble solids (TSS) content, pH, titratable acidity, betalains content, total soluble phenols (TSP) content, and antioxidant activity.
Physical and physiological variables
Weight was assessed with a digital scale (Ohaus, USA) with precision of 0.1 g. Cumulative weight loss was evaluated in percentage (% WL) based on the initial condition. Color was measured in fruit skin and fruit flesh with a Hunter Lab colorimeter (Mini Scan XE Plus 45/0-L, USA) and was expressed in hue angle (h°), chromaticity (C*), and lightness (L*) (McGuire, 1992).
The respiration rate was evaluated with a static method (Hernández-Muñoz, Almenar, del Valle, Velez, & Gavara, 2008). The
experimental unit was placed in an airtight container for 45 min and the change
in CO2 concentration 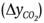 was determined. To do
this, gaseous samples of 3 mL were obtained from the headspace of recipients and
they were placed in glass tubes of 7 mL with 4 mL of a sodium bicarbonate
solution containing bromothymol blue as indicator. Agitation was applied for 15
s and absorbance was measured with a spectrophotometer (Perkin Elmer Lambda 25
UV/Vis, USA) at 615 nm. CO2 mixtures with concentration in the range
of 0.2 to 12.5 % were prepared in order to construct a standard curve, which was
used to quantify the concentration of CO2 in the headspace of samples
(García-Cruz et al., 2016). Then,
based on fruit weight (mfr), container
free volume (VL), and elapsed time
(Δt = 45 min = 0.75 h), respiration rate
(R) was determined in mL⋅kg-1⋅h-1
with the calculation:
was determined. To do
this, gaseous samples of 3 mL were obtained from the headspace of recipients and
they were placed in glass tubes of 7 mL with 4 mL of a sodium bicarbonate
solution containing bromothymol blue as indicator. Agitation was applied for 15
s and absorbance was measured with a spectrophotometer (Perkin Elmer Lambda 25
UV/Vis, USA) at 615 nm. CO2 mixtures with concentration in the range
of 0.2 to 12.5 % were prepared in order to construct a standard curve, which was
used to quantify the concentration of CO2 in the headspace of samples
(García-Cruz et al., 2016). Then,
based on fruit weight (mfr), container
free volume (VL), and elapsed time
(Δt = 45 min = 0.75 h), respiration rate
(R) was determined in mL⋅kg-1⋅h-1
with the calculation: 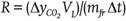 .
.
Firmness was evaluated using a texture analyzer (TA-XT2i, Stable Micro Systems, UK) at two points of the equatorial region of fruit, with a routine that consisted of applying a compression force with a spherical probe of 10 mm in diameter that deformed tissue to a depth of 5 mm at a velocity of 5 mm⋅s-1. Additionally, a microphotograph of the epidermal section of fruit was obtained. Samples of the epicarpic region were crushed and fixed with 2.5 % glutaraldehyde for 24 h. Three washes with a phosphate buffer were applied, and materials were then sequentially suspended in ethanol solutions with concentrations from 50 to 100 % (v/v) and subsequently dried. A fine layer of gold was applied using a sputter coater (JFC-1100 Fine Coat; JEOL LTD, Japan). Finally, a scanning electron microscope (JSM-6390; JEOL Ltd., Japan) was used to obtain images of the cross section of samples.
Chemical variables
Total soluble solids (TSS) were expressed in °Brix and were measured with a Master-M refractometer (Atago®, Japan) in a drop of juice taken from fruit flesh. To evaluate pH and titratable acidity, 5 g of fruit flesh were macerated with 50 mL of distilled water. Filtration with cheesecloth was applied and the liquid was first analyzed with a potentiometer (Hanna Instruments, Romania) to determine pH and thereafter it was titrated with NaOH 0.01 N to determine titratable acidity (Horwitz, 1980).
Betalains and total soluble phenols content
The method of Wu et al. (2006) was used to obtain a methanolic extract from fruit. Two grams of flesh were macerated with 20 mL of methanol 80 % (v/v). Sonication was applied for 10 min with a Branson® (USA) bath. Mixtures were then shaken in dark for 20 min at room temperature and centrifuged at 2,200 x g on a Hettich Zentrifugen unit (Model Universal 32, Germany). The supernatant was separated and the residue was subjected to a similar second extraction. The supernatants were pooled, filtered with No. 4 Whatman paper, and concentrated to dryness at 40 °C on a rotary evaporator (Laborata 4010, Germany). Finally, residues were re-suspended in 10 mL of a solution of methanol 80 % and they were stored in amber containers at -20 °C.
Betacyanin and betaxanthin concentrations were determined with the method of Castellanos-Santiago and Yahia (2008) through spectrophotometry and the calculation: B = (A x DF x W x V) / (Ɛ x P x L), where B is content of betacyanins or betaxanthins (mg⋅g-1), A is absorbance (538 nm for betacyanins and 483 nm for betaxanthins), DF is a dilution factor when reading was made, W is molecular weight (550 g⋅mol-1 for betanin and 308 g⋅mol-1 for indicaxanthin), Ɛ is the molar extinction coefficient (60,000 L⋅mol-1⋅cm-1 for betanin and 48,000 L⋅mol-1⋅cm-1 for indicaxanthin), P is mass of sample (g), and L is the length (1 cm) of the cuvette used during the determination. Results were expressed as total content of betalains per 100 g of fresh weight, through the sum of the contents of betacyanins and betaxanthins.
The determination of TSP was carried out in the same methanolic extract, using the Folin-Ciocalteu method (Singleton & Rossi, 1965). 100 μL of the extract reacted with 125 μL of the Folin-Ciocalteu reagent for 6 min. Then neutralization was applied with 1,250 μL of a solution of Na2CO3 (19 %) and the volume was adjusted to 3 mL with distilled water. Mixtures were shaken on a vortex and placed in darkness for 90 min to achieve stabilization. Centrifugation (Hermle Z200 equipment, Labortechnik, Germany) was applied at 15,300 x g for 10 min and absorbance (Perkin Elmer Lambda 25 UV/Vis, USA) was measured at 760 nm. A standard curve with gallic acid was prepared to express TSP content as mg of gallic acid equivalents per kilogram of fresh weight (mg GAE⋅kg-1).
Antioxidant activity
A 7 mM solution of ABTS [2,2’-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid)] was mixed with 2.45 mM of potassium persulfate (K2S2O8; PP) at a 2:1 ratio. The mixture was placed in darkness for 16 h to allow free radical generation and afterwards diluted with a phosphate buffer at pH 7.4 until absorbance was 0.7 at 734 nm (Wu et al., 2006). Aliquots of 200 μL of the methanolic extract reacted with 2,800 μL of the ABTS-PP solution and absorbance was measured every minute for 7 min. The reduced ABTS was determined with the calculation: %ABTS = (A0 - An)100/A0 , where A0 and An were blank and sample absorbances, respectively. Trolox [(±)-6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid] at different concentrations (50-300 μmol⋅L-1) was used to prepare a standard curve and antioxidant capacity was expressed in μmol of Trolox equivalents per kilogram of sample (μmol TE⋅kg-1).
Data analysis
The work was conducted according to a 2×2 factorial arrangement in a completely randomized design. Variation factors were fruit variant (with red or orange flesh) and the presence or absence of thorns. In addition, the significance of changes over the storage time was evaluated. An analysis of variance, complemented with means comparison routines (Tukey, P ≤ 0.05), was carried out to analyze data.
Results and discussion
Physical and physiological variables
Fruit weight ranged from 168.1 to 197.6 g and during storage showed continuous mass loss that occurred at a rate of 0.55, 0.86, 0.46, and 0.82 % per day in the RwT, RoT, OwT, and OoT treatments, respectively (Figure 2A). Transpiration is a phenomenon resulting from the presence of a water vapor deficit (Maguire, Banks, & Opara, 2001) and in this case it was caused by 76 % relative humidity, which led to water transport from within the product to the surrounding air. This phenomenon was similar in both variants of fruit, red and orange, but it was significantly affected by thorn removal (P ≤ 0.05; Table 1), since the highest losses occurred in fruit without thorns.
Table 1..
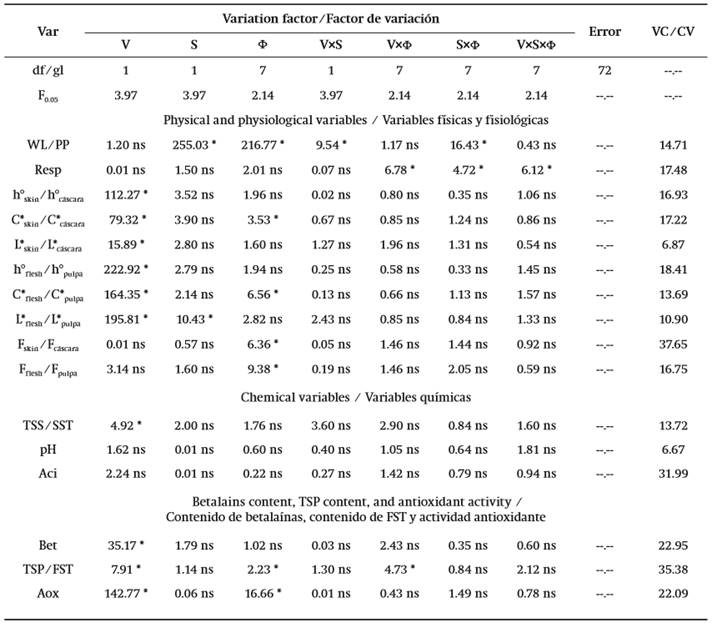
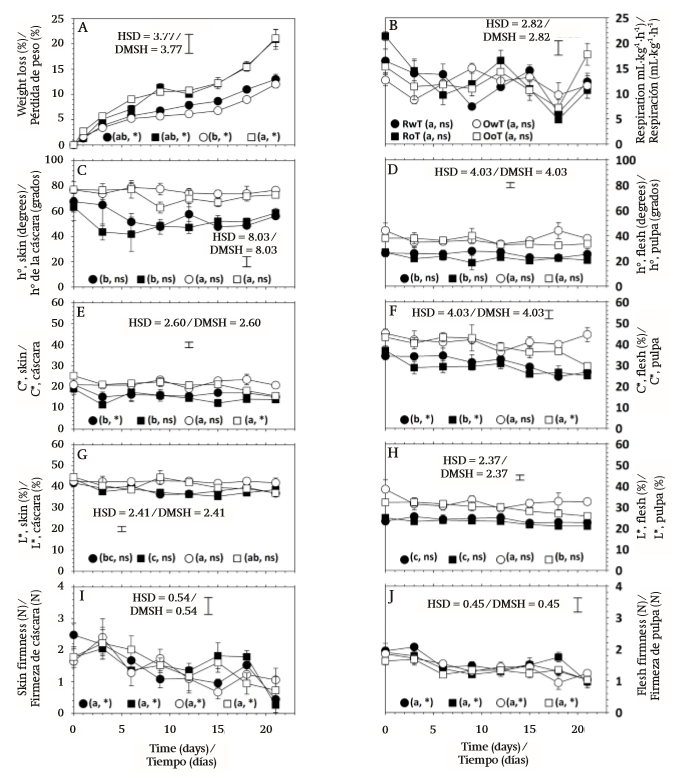
The epicarp of S. pruinosus fruit is approximately 0.61 cm thick and possesses areoles ranging in number from 24 to 28 (García-Cruz et al., 2016). The most external region consists of a continuous cuticle and through microphotography the presence of waxes on the surface could be detected (Figure 3), which suggests that transpiration occurs mainly in the areole points, where cuticle is interrupted and, when thorns are removed, the contact area with the external environment is increased, causing an increment in the transpiration rate, which enhances mass loss by up to 43.8 % in red fruit and up to 75.5 % in orange fruit. On the other hand, the behavior over time was similar between fruit without thorns, but it contrasted slightly between fruit variants with thorns, which explained the significance of the V×S and S×F interactions (Table 1).

The respiration rate was not affected by fruit variant (Table 1). During storage there were fluctuations (Figure 2B) resulting from the fact that different experimental units were used at different temporal sampling points. However, such changes over time were not significant (P > 0.05; Table 1). According to Armella, Yánez-López, Soriano, and Ramírez (2003), the fruit of the Stenocereus genus exhibit a non-climacteric behavior and respiration activity occurs without significant modifications during ripening and also after consumption maturity is reached.
On the other hand, although major effects were nonsignificant, the statistical analysis reported that two double interactions were significant and, among all variables, respiration was the only case where the triple interaction was significant (Table 1). The respiratory activity in the present work was evaluated at 12 °C and had average values of 11.99 (± 3.59), 12.55 (± 4.82), 11.90 (± 2.28), and 12.41 (± 3.44) mL∙kg-1∙h-1 in RwT, RoT, OwT, and OoT, respectively. This suggests that thorn removal caused a slight increment in metabolic activity. Even though such anatomical elements are deciduous and their separation does not require any mechanical effort, the presence of greater open areas could favor gaseous exchange with the environment, resulting in a higher respiration rate, similarly to what occurred with the weight loss. However, variability in measurements was high and the significance in interactions was in fact due to contrasts that occurred in several days during storage, but without any clear tendency (Figure 2B). On the other hand, García-Cruz et al. (2016) reported values of respiratory activity at 24 °C of 9.5 μg∙kg-1∙s-1 (24.71 mL∙kg-1∙h-1) and 11.1 μg∙kg-1∙s-1 (28.79 mL∙kg-1∙h-1) for fruit of S. pruinosus with red and orange flesh, respectively, which implies that a Q10 value equal to 1.83 and 2.09 characterized the dependence of respiration with temperature in the range from 12 to 24 °C for red and orange fruit, respectively.
Color is a distinctive feature of S. pruinosus fruit variants and, similarly to what was reported by García-Cruz et al. (2016), there was significant difference in hue angle (h°) and chroma (C*) between red and orange fruit, both in skin and flesh (Table 1). Hue angle had values between red and yellow tones in the CieLab space (McGuire, 1992). In the case of skin, average values, in degrees, were 55.22 (± 13.49), 50.78 (± 12.24), 75.72 (± 5.86), and 71.93 (± 8.15) for RwT, RoT, OwT, and OoT, respectively, which means that thorn removal caused a reduction of 8.04 and 5.01 % in the hue angle of red and orange fruit, respectively, although the statistical analysis reported that the contrast was non-significant (Table 1, Figure 2C). Hue angle of skin contrasted with that of flesh, where average values, in degrees, were 25.37 (± 5.16), 22.08 (± 4.35), 37.67 (± 6.40), and 35.50 (± 4.78), for the same treatments, respectively, and where the effect of thorn removal was again non-significant (Table 1, Figure 2D).
In the case of C*, average values were 16.79 (± 3.89), 14.97 (± 3.84), 21.61 (± 2.41), and 20.86 (± 3.12) in skin, and 30.91 (± 5.45), 29.05 (± 4.62), 41.46 (± 4.72), and 38.88 (± 5.99) in flesh, for fruit of RwT, RoT, OwT, and OoT, respectively (Figure 2E and 2F), which showed that the thorn removal caused a 7.16 % loss of chroma in skin and 6.12 % in flesh, although the statistical analysis report, as occurred before, indicated that contrasts were non-significant (Table 1).
On the other hand, the storage time did not affect the hue angle of skin or flesh, suggesting that the composition of betalains, which are compounds that provide the color to these fruit (García-Cruz et al., 2013), did not change during storage. In contrast, chroma was modified over time (Table 1), especially in the case of flesh, where a reduction of 0.46 units per day was observed in red fruit and of 0.33 units per day in orange fruit (Figure 2F), and this differential loss of C* explained the significant interaction that was found in the analysis of variance between factors of fruit variant and storage time (Table 1). Additionally, such interaction was also significant in the case of chroma of skin, but in that case this was explained because the time only affected treatments RwT and OoT (Figure 2E), although the observed modifications had no practical importance.
Lightness (L*) was affected by the type of fruit and, similarly to the above attributes, there was contrast between skin and flesh (Figure 2G and 2H). Average values were 38.69 (± 2.92), 38.06 (± 3.04), 42.47 (± 2.01), and 40.86 (± 3.86) % in skin and 23.83 (± 2.08), 22.82 (± 1.90), 32.58 (± 4.61), and 29.68 (± 2.91) % in flesh, for RwT, RoT, OwT, and OoT, respectively. In the case of flesh there was significant contrast in this attribute between red and orange fruit (Figure 2H). However, although the statistical analysis also reported significant difference in the case of skin, such contrast was actually little evident (Figure 2G). In the case of flesh, additionally to the effect given by the type of fruit, it was found that the action of removing thorns caused a significant modification of lightness (Table 1), but when means were compared this was only confirmed for orange fruit (Figure 2H), where L* diminished by 8.90 %.
Firmness of fruit was less than 2 N and this was similar to values reported by García-Cruz et al. (2016). The pitaya fruit have consistency that is perceived as very soft when they are touched (data not shown) and the firmness values found confirmed that they are very fragile materials, which makes them susceptible to mechanical damage, especially for the puncturing action of thorns, and this should be taken into account in the design of packaging for postharvest handling.
Firmness was not affected by the type of fruit or by the removal of thorns (P > 0.05), but it was affected by storage time (Table 1), because this mechanical attribute decreased as days passed (Figures 2I and 2J), at rates of 0.083, 0.039, 0.044, and 0.054 N per day in skin, and 0.040, 0.028, 0.035, and 0.024 N per day in flesh of fruit of treatments RwT, RoT, OwT, and OoT, respectively. This phenomenon of loss of firmness after harvest has been observed in other cactaceae fruit like prickly pear (Ochoa-Velasco & Guerrero-Beltrán, 2014) and those of Hylocereus undatus (Osuna-Enciso et al., 2011; Zahid, Ali, Siddiqui, & Maqbool, 2013).
Fruit softening is associated with degradation of cell wall components through enzymatic mechanisms, but also with the turgor loss caused by a transpiration phenomenon (Brummell, 2006; Smith, Waldron, Maness, & Perkins-Veazie, 2003). In the case of pitaya, due to a non-climacteric behavior (Armella et al., 2003), fruit are harvested at consumption maturity, when changes of firmness associated with ripening (Paul, Pandey, & Srivastava, 2012) have already occurred. However, the behavior found in the present work suggests that the loss of consistency is a phenomenon that continues even in the senescence phase.
Chemical variables
Total soluble solids (TSS) were only affected by the type of fruit, red or orange, as there was no significant effect derived from the removal of thorns or storage time (Table 1), and this was attributed to the non-climacteric behavior of fruit (Reid, 2002). The highest values were found in orange fruit, with 11.31 (± 2.07) °Brix in OwT and 11.46 (± 2.09) °Brix in OoT, and the lowest in red fruit, with 10.69 (± 1.25) °Brix in RwT and 9.69 (± 1.40) °Brix in RoT (Figure 4A). In general, the content was similar to that reported by García-Cruz et al. (2016) for this same species.
In contrast to this behavior, pH and titratable acidity were not affected by any of the variation factors, fruit type, thorn removal or storage time (P > 0.05; Table 1). Average values of pH were 5.55 (± 0.40), 5.50 (± 0.36), 5.60 (± 0.35), and 5.65 (± 0.31), for treatments RwT, RoT, OwT, and OoT, respectively, while those of titratable acidity (TA) were 0.081 (± 0.023), 0.084 (± 0.020), 0.074 (± 0.028), and 0.073 (± 0.025), respectively (Figures 4B and 4C).
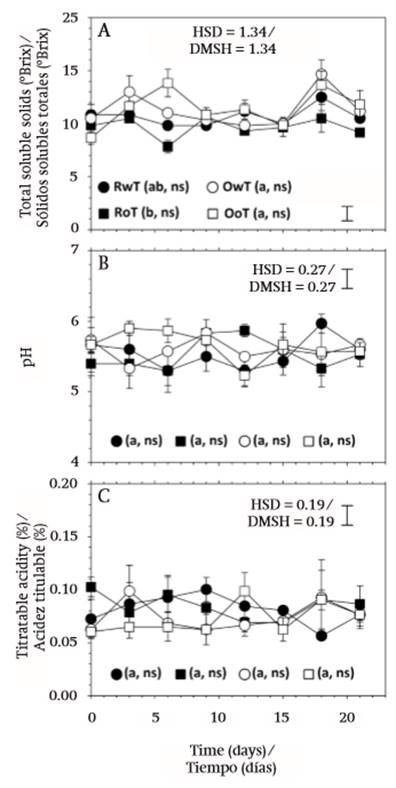
According to Famiani, Battistelli, Moscatello, Cruz-Castillo, and Walker (2015), the most common acids in fleshy fruits are citric and malic. In the case of pitaya fruit, the acids that impart sourness have not been determined yet. However, the content found contrasted with that of other fruit of the Stenocereus genus, like S. stellatus (García-Cruz et al., 2016) or that of other cactaceae fruit like pitahaya (Hylocereus spp.; Rodríguez-Rodríguez, Patiño-Gutiérrez, Miranda-Lasprilla, Fisher, & Galvis-Venegas, 2005; Esquivel, Stintzing, & Carle, 2007). The acid content and that of sugars play an important role in fruit flavor (Famiani et al., 2015). In the case of fruit of S. pruinosus, these are recognized by a sweet flavor, given by a high TSS/TA ratio, which in the present work varied between 115.5 and 132.0 in red fruit and between 152.8 and 157.0 in orange fruit.
Contents of betalains, total soluble phenols, and antioxidant activity
The color exhibited by pitaya fruit is due to the presence of betalains (García-Cruz et al., 2013), which varied in content from 4.51 to 4.87 mg∙kg-1 in red fruit and from 2.35 to 2.63 mg∙kg-1 in orange fruit (Figure 5A). The difference between both types of fruit was significant (Table 1) and this was coherent with the contrast observed in tonality or hue angle of flesh and with the report of García-Cruz et al. (2016), who determined that in red fruit there were five betalains present, but only four of them were identified in orange fruit. In addition, there was no significant effect caused by thorn removal and, although in orange fruit a reduction during storage in the betalains content was visually observed in graphs (Figure 5A), the statistical analysis did not confirm the significance of the modification (Table 1). On the other hand, that lack of significance in the effect of storage time on betalains content explained the constancy of the hue angle (h°) over time (Figures 2C and 2D).
Pitaya fruit with red and orange flesh also showed different content of total soluble phenols (TSP) and this characteristic was not affected by thorn removal (Table 1). The average values of TSP content were 459.36 (± 87.79), 386.61 (± 92.75), 220.43 (± 36.15), and 222.76 (± 79.66) mg∙kg-1 in fruit of RwT, RoT, OwT, and OoT, respectively, and were similar to those obtained by García-Cruz et al. (2016), who also reported the contrast between fruit with red and orange flesh. On the other hand, TSP content diminished during storage, at a rate of 13.86, 11.55, 5.92, and 10.08 mg∙kg-1 per day, respectively (Figure 5B), and these results contrasted with those reported by García-Cruz et al. (2016) in the sense that they observed that such compounds remained constant in content during storage, but in fact the period that they evaluated was only of 6 d at 24 °C.

The reduction of phenolic compounds in fruit has been commonly associated with the activity of enzymes like polyphenol oxidase and peroxidase (Tomás-Barberán & Espín, 2001). However, studies about the physiology and biochemistry of pitaya fruit are scarce and clarification of certain aspects related to biosynthesis and degradation of phenolic compounds still requires the development of additional research.
It is documented that both betalains and phenolic compounds exhibit antioxidant activity (Kaur & Kapoor, 2001; Azeredo, 2009) and, due to this, pitaya fruit can be considered as nutraceutical food (Wang, Melnyk, Tsao, & Marcone, 2011), which can increase the commercialization value in the fresh fruit market. In this regard, the antioxidant activity had average values of 4.69 (± 0.72), 4.64 (± 0.52), 2.84 (± 0.36), and 2.77 (± 0.50) μmol TE∙kg-1 in fruit of RwT, RoT, OwT, and OoT, respectively. The difference between red and orange fruit was significant (P ≤ 0.05) but there was no important effect derived from thorn removal (Table 1).
On the other hand, antioxidant activity values remained largely constant during the first 14 d, but thereafter this attribute experienced a reduction of 26.54, 33.62, 40.89, and 20.04 % in RwT, RoT, OwT, and OoT, respectively (Figure 5C), when day 21 was reached. García-Cruz et al. (2016) suggested that the antioxidant potential of pitaya fruit is provided more by phenolic compounds and to a lesser extent by betalains. In the present work betalains content remained constant during storage, but TSP diminished, which strengthens the argument that the antioxidant activity is mostly provided by phenolic compounds.
Shelf life
Shelf life is the period of time over which a food retains attributes at an acceptable level for consumption, considering nutritional aspects, sensory perception, physical and chemical properties, and those that affect safety (Hough, 2010; Institute of Food Science Technology [IFST], 1993). García-Cruz et al. (2016) determined the shelf life of Stenocereus pruinosus and Stenocereus stellatus fruit maintained at 24 °C to be about six days, because after that time fungal growth occurred. In the present work no microbial contamination was observed during storage and most quality attributes remained at approximately constant values derived from the non-climacteric nature of fruit (Armella et al., 2003). However, from day 12 of storage the weight loss rate increased in fruit where thorns were removed (Figure 2A). In addition, from day 14 the antioxidant capacity began to decline (Figure 5C) and, therefore, the nutraceutical potential of fruit (Wang et al. 2011) also diminished.
In this regard, Andersson et al. (2015) pointed out that treatments focused on shelf life should take into account the preservation of bioactive compounds. Moreover, firmness in skin acquired values below 1 N after 12 days of storage (Figure 2I), and this phenomenon made fruit too fragile for handling in fresh condition. Therefore, based on these results, the handling period of pitaya fruit should not exceed 12-14 days at 12 °C after harvest. However, it should also be considered that in the present work a treatment with NaClO was applied before storage in order to reduce microbial load, and this might have contributed to the lengthening of shelf life. On the other hand, since thorn removal had a negligible impact on most quality attributes, it is advisable to carry out this practice immediately after harvest, in order to reduce the risk of mechanical damage by puncturing fruit.
Conclusions
Handling at 12 °C caused a reduction in the metabolic activity of pitaya fruit, allowing lengthening of shelf life to a period between 12 and 14 days. The removal of thorns from fruit affected only the fruit weight loss rate and loss of lightness in skin, so it was considered an advisable practice. Based on the nutraceutical potential derived from antioxidant activity, consumption of fruit of Stenocereus pruinosus in fresh state is recommended.
