Introduction
In Mexico, the Bradysia impatiens Johannsen (Diptera: Sciaridae) fungus gnats is considered a pest with forest and agricultural importance for nurseries and greenhouses in the center of the country, especially for the production of seedlings (Cibrián, García, & Don Juan, 2008; Marín-Cruz, Cibrián-Tovar, Méndez-Montiel, Pérez-Vera, & Cadena-Meneses, 2015a). Bradysia impatiens larvae feed on decaying organic matter, fungi and roots (Mohrig & Menzel, 2009). The root, being damaged, is exposed to the entry of pathogenic fungi, and larvae may carry spores of them (Shamshad, Clift, & Mansfield, 2009).
It has been reported that B. impatiens damages roots and bulbs, mainly in greenhouses and nurseries, ornamental, from vegetables and in gardens (Marín-Cruz et al., 2015a). The control is carried out by means of organosynthetic insecticides, growth regulators and biopesticides; however, some populations of B. impatiens are resistant to chemical treatments (Marín-Cruz et al., 2015a). An alternative is the use of entomopathogenic fungi (EF), which are found in the environment and have potential as insect controllers, causing fungal infections in arthropods (Motta-Delgado & Murcia-Ordoñez, 2011).
Beauveria bassiana (Balsamo) Vuillemin (Ascomycota: Hypocreales) is an EF with cosmopolitan distribution that attacks different orders of insects (Lozano-Tovar, Ortiz-Urquiza, Garrido-Jurado, Trapero-Casa, & Quesada-Moraga, 2013; Zibaee, Bandani, Talaei-Hassanlouei, & Malagoli, 2011).The fungus infects insects from the order Diptera and has effective control of Musca domestica Linnaeus (Muscidae), Ceratitis capitata Wiedemann (Tephritidae) and Aedes aegypti Linnaeus (Culicidae) (Acharya, Seliga, Rojotte, Jenkins, & Matthew, 2015; Lozano-Tovar et al., 2013).
The fungus B. bassiana produces secondary metabolites that have insecticidal properties and important applications in biotechnology (Sánchez-Pérez, Barranco-Florido, Rodríguez-Navarro, Cervantes-Mayagoitia, & Ramos-López, 2014), including bassianolides, bassiacriin, bassianin, beauvericin, beauverolides and tenellin (Quesada-Moraga & Vey, 2004). Bassianolide affects populations of Lepidoptera such as Galleria mellonella Linnaeus (Pyralidae), Helicoverpa zea Boddie (Noctuidae) and Spodoptera exigua Hübner (Noctuidae) (Xu et al., 2009). The bassiacridin injected into Locusta migrant Linnaeus (Orthoptera: Acrididae) causes alterations in the epithelial cells of the trachea and displacement of the integument (Quesada-Moraga & Vey, 2004). Secondary metabolites of B. bassiana disrupt the mechanisms of the insect's immune system, allowing the fungus to invade and kill its host (Zibaee et al., 2011).
The present study evaluated the insecticidal and insectistatic potential of strain 11 B. bassiana using conidia, crude extract (CE) and lyophilized (enzymes and metabolites) in third instar larvae of B. impatiens.
Materials and methods
Biological material
Collection and breeding of B. impatiens insects
In the forest nursery of Temamatla, Estado de México, larvae and adult B. impatiens were collected from 200 seedlings of Pinus montezumae (Lamb) (Pinales: Pinaceae) during November 2014. B. impatiens adults were collected using a bee vacuum pump and larvae were collected with a brush directly from the root and growth substrate of the pine.
The insects collected were taken to the insectarium laboratory of the Universidad Autónoma Metropolitana, Unidad Xochimilco (UAM-X), and were maintained under photoperiod conditions at 12:12 h, relative humidity (RH) of 75 %, temperature 23 ± 2 °C, with 2 g of potato slices as food (Marín-Cruz et al., 2015b).
Activation of B. bassiana
Strain 11 B. bassiana from the biotechnology laboratory of UAM-X was grown on Dextrose-Saboraud agar for 15 days at 25 °C. Conidia were harvested with 0.05 % of Tween 80 by means of gentle scraping and were counted with a hemocytometer; the concentration was adjusted to 107 conidia·mL-1 (Barranco et al., 2009).
Conidia and crude enzymatic extract of B. bassiana
A solid state medium consisting of shrimp shell, sugar cane bagasse, mineral salts (NH4)2SO4, MgSO4, NaCl, KH2PO4, FeSO4·7H2O, ZnSO4 and MnSO4 was prepared using Erlenmeyer flasks (Barranco et al., 2009). B. bassiana EF was inoculated at a concentration of 107 conidia·mL-1. Growing conditions were pH 5.0 and 25 °C (Barranco-Florido, Alatorre-Rosas, Gutiérrez-Rojas, Viniegra-González, & Saucedo-Castañeda, 2002). Conidia were harvested at 15 days using 0.05% of Tween 80 1:10 (v / v), the count was carried out using a hemocytometer and the concentration was adjusted to 107 conidia·mL-1. The enzymatic crude extract was obtained at 5 days with distilled water in a ratio of 1:1 (v/v) (Barranco-Florido et al., 2002).
Semipurification of crude enzymatic extract
The crude extract was centrifuged at 10 000 rpm for 10 min (Sorvall® model RC-5B, USA). Subsequently a second centrifugation was carried out to separate the molecular size from the supernatant (BECKMAN®, GPR model, USA) using 10 kDa Centricon tubes (BioRad®, Ireland) at 3 000 rpm for 30 min (Ali, Ren, Huang, & Wu, 2010). The upper extract, greater than 10 kDa, corresponds to the enzymes; and the lower, less than 10 kDa, to metabolites. For application in the treatments, metabolites and enzymes were lyophilized (Labconco®, model 7753022, USA) and resuspended in distilled water at a dose of 10 000 ppm.
Bioassay of insecticidal and insectistatic activity of B. bassiana
Products from the purification of enzymatic extract and conidia of B. bassiana were evaluated in third instar B. impatiens larvae, deposited in transparent plastic boxes (1.5 cm diameter) with cover, to avoid dehydration; a piece of 1 x 1 cm adsorbent paper containing the treatments was place in each box. The larvae were fed with 20 μg of potato flour, used for the consumption of treatments (Table 1). The dose was a single application of 130 μL of each treatment and distilled water was applied to the control treatment. As for the conditions, a temperature of 23 ± 3 °C was maintained and RH of 60 to 70 %. A total of 20 μL of distilled water was added to the plastic boxes every 72 h to conserve moisture, with checks every 24 hours. The experimental design was completely randomized with five replications per treatment; each replication consisted of 10 larvae, using 50 larvae in total per treatment.
Table 1.
| Treatment | Dose on adsorbent paper (µL) | Dose in food (µg·µL-1) |
|---|---|---|
| Conidia (107 conidia·mL-1) | 130 | 5.7 |
| Crude extract | 130 | 5.7 |
| Enzymes (10 000 ppm) | 130 | 5.7 |
| Metabolites (10 000 ppm) | 130 | 5.7 |
| Control | 130 | 5.7 |
Evaluated variables
Insecticidal activity (mortality) of B. bassiana
The number of dead larvae and pupae was recorded every 24 h for eight days; with these data mortality corrected by means of the Abbott equation (1925) was calculated to eliminate the natural mortality obtained in the control:
Insectistatic activity of B. bassiana
The criteria for defining insectistatic activity were: 1) duration of B. impatiens pupa, 2) relative emergence of adults from pupa, and 3) morphological alterations in larvae, pupae and adults. Revisions were made every 24 h for 20 days, and relative emergence was calculated using the following formula (Aguilera, 2001):
Statistical analysis
The percentage of corrected mortality and relative emergence had a binomial distribution; to normalize them we used the formula of angular or arcsine transformation (
Results and Discussion
Insecticidal activity of B. bassiana
Table 2 shows the results of corrected mortality of B. impatiens (larvae and pupae) (F 3,16 = 311.58, P ˂ 0.001). Figure 1 shows that conidia and crude extract treatments had an effect on the mortality rate after 48 h, while metabolites and enzymes showed effect until the sixth and eighth day, respectively.
Table 2.Corrected.
| Period (days) | Treatment | Corrected mortality (%) | Arcsine transformation |
|---|---|---|---|
| 8 | Control | 0.0 | 0.0d |
| Metabolites | 15.5 | 23.22c | |
| Enzymes | 0.0 | 0.0d | |
| Conidia | 31.1 | 33.90a | |
| Crude extract | 22.2 | 28.12b | |
| 20 | Control | 0.0 | 0.0d |
| Metabolites | 47.5 | 43.56a | |
| Enzymes | 4.4 | 12.16c | |
| Conidia | 42.1 | 40.45a | |
| Crude extract | 22.2 | 28.12b |
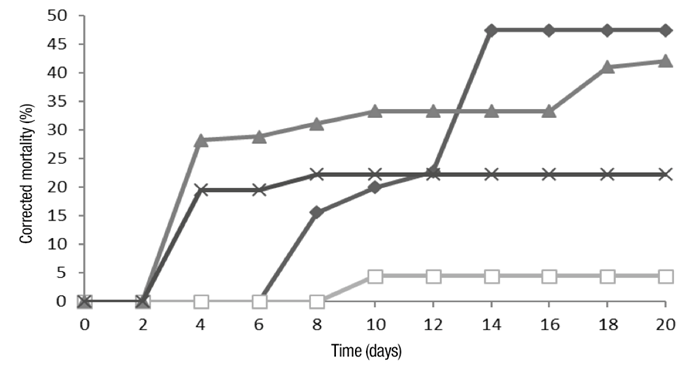
According to the results, at eight days, mortality is less than 40 % in all treatments, therefore, insecticidal activity is considered reduced. The effect of treatments at 20 days was statistically different (F 3,16 = 73.02, P ˂ 0.001). From day 8 to 20, metabolites significantly (31.95 units) increased mortality compared to conidia (10.99 units) (Table 2). Crude extract did not increase mortality in that period (Figure 1).
The insecticidal activity of B. bassiana, per contact, has been demonstrated in larvae of the orders Coleoptera and Lepidoptera (Ahmed & El-Katatny, 2007; Guzmán-Franco et al., 2012; Michelle et al., 2013). Also Acharya et al. (2015) evaluated the insecticidal activity of B. bassiana in adults of M. domestica (Diptera) and reported 100% of mortality in a period of six to 10 days after application. There is few information regarding the ingestion of B. bassiana conidia by diptera larvae.
Insectistatic activity of B. bassiana
Metabolites did not cause mortality of B. impatiens larvae in the first six days, but produce mortality in the following days. This may be because toxicity was cumulative, due to feeding, causing death. It should be noted that after 14 days mortality ceases, but the effect of metabolites continues at physiological level interfering in the development of B. impatiens, which is denominated insectistatic effect.
The insectistatic effect is manifested in the inhibition of feeding, growth, development and oviposition of the insect (Rodríguez, 2003). In our study, low molecular weight metabolites had higher insectistatic effect than insecticidal activity. The insectistatic effect was demonstrated with the inhibition of the growth of larvae, causing their death.
Pupae of B. impatiens
Figure 2 shows the percentage of larvae that passed to the pupal stage in the different treatments, with significant difference among them (F 4,20 = 175.32, P ˂ 0.001). As effect of the insectistatic activity, mortality of the pupae in treatments was: control 12 % (5); Low molecular metabolites 44 % (16); Enzymes 8 % (6); Conidia 71 % (5) and CE 14 % (6). The number of dead pupae and malformed adults is evidence of inhibition of development as a result of insectistatic activity, and may be related to juvenile hormones, which cause insect larvae to continue molting. If the production of these hormones ceases during the last larval or nymphal stage, then transformation occurs to pupal stage and later to adult (Rodríguez, 2003). The insectistatic effect of metabolites of EF has not been demonstrated, only the effect of extracts of plants have been mentioned, where the main result is the extension of the larval and pupal stages of Spodoptera frugiperda Walker (Lepidoptera: Noctuidae) (Zavala-Sánchez, Pérez-Gutiérrez, Romo-Asunción, Cárdenas-Ortega, & Ramos-López, 2013).

Moreover, it was observed that, at the end of the treatment with metabolites, there was no presence of B. impatiens larvae and pupae. The results obtained in this study are similar to those reported by Lozano-Tovar et al. (2013), who obtained 45 % of maximum mortality caused by B. bassiana in pupae of C. capitata.
Emergence of adults of B. impatiens
Table 3 shows the relative emergence results for adults at 8 and 20 days (F 4,20 = 158.70, P ˂ 0.001). Of the 20 adults who emerged in the treatment with low molecular metabolites, 13 (65 %) showed malformations, representing 26 % of the total of 50 treated insects; in addition, only seven adults, 14 % of the total larvae, emerged with reproduction capacity.
Table 3.
| Period (days) | Treatment | Adults (%) | Relative emergence (%) | Arcsine transformation |
|---|---|---|---|---|
| 8 | Control | 46 | 100 | 90a |
| Metabolites | 13 | 28 | 32b | |
| Enzymes | 46 | 100 | 90a | |
| Conidia | 0 | 0 | 0d | |
| Crude extract | 2 | 4 | 12c | |
| 20 | Control | 67 | 100 | 90a |
| Metabolites | 40 | 60 | 51b | |
| Enzymes | 67 | 100 | 90a | |
| Conidia | 4 | 6 | 14c | |
| Crude extract | 38 | 57 | 49b |
The emergence of malformed adults reduces the number of adults adequate for reproduction; this symptom is consistent with that reported by Abd El-Ghany, El-Sheikh, Abd El-Rahman, and Abd El-Nasser (2012), who observed malformations in adults of G. mellonella after application of crude extract of B. bassiana. These malformations may be caused by cicliyc depsipeptides nature called beauvericin. This toxin also produces alterations in the processes of moulting and metamorphosis (Ferron, 1978).
The insecticidal activity of the crude extracts of EF has been evaluated in several genera of insects, mainly in the orders Coleoptera and Lepidoptera (Guzmán-Franco et al. 2012; Zibaee et al., 2011). Ortiz-Urquiza, Garrido-Jurado, Santiago-Álvarez, and Quesada-Moraga (2009) had 80 % of mortality five days after the application of crude enzymatic extract of Metarhizium anisopliae (Mestch) in the adult food of C. capitata. Regarding the purified metabolites, Quesada-Moraga and Vey (2004) reported the insecticidal effect of bassiacridin on L. migratoria; however, in this study there was lower mortality in the treatments with enzymes and crude extract, compared to those obtained by these authors. This can be attributed to the survival capacity of B. impatiens (Marín-Cruz et al., 2015b), as dipterans have evolved into niches where pathogens, such as fungi and bacteria, abound (Kuraishi, Hori, & Kurata, 2013). In addition to the above, dipterans have defense mechanisms such as physical barriers (peritrophic membrane type II) and regeneration of damaged cells in the epithelium, which are very efficient in detecting and counteracting the effect of foreign agents (Kuraishi et al., 2013).
Symptomatology of B. impatiens treated with extracts of B. bassiana
Due to treatments, dead larvae showed rigidity and melanization in all the treatments, in contrast to dead larvae of the control, which were flaccid and kept a light color. This symptomatology agrees with what has been pointed out by other authors. Chávez, Rodríguez, Sánchez, Hamdan, and Barranco (2014) reported that the application of crude extract of B. bassiana in larvae of Phyllophaga spp. Harris (Coleoptera: Melolonthidae) causes melanization of them self. Melanization is a response process from the insect humoral immune system to the presence of some external agent (González-Sántoyo & Córdoba-Aguilar, 2012); the change of color, characteristic of the B. impatiens pupae, may be due to this process. The insectistatic effect is manifested by the prolongation of the time that a larva or pupa remains in a stage; in the case of B. impatiens pupae, this effect has an average duration of four days (Marín-Cruz et al. 2015b). In the present study, pupae from which no adult emerged, after seven days, began to decompose and become dark in color. The treatment of conidia generated mycelium of B. bassiana in some cases. Dead larvae due to low molecular weight metabolites were rigid; in addition, 13 adults had malformations in the wings that prevented them to spread their wings and flying. In the case of malformed pupae there was no adult emergence. This insectistatic effect is important because, although it does not have an immediate effect or in the first days, its action of inhibiting the growth and normal development of larvae and pupae of B. impatiens causes that they do not complete their cycle.
Conclusions
Conidia and extracts of Beauveria bassiana (enzymes, metabolites and crude extract) had different effects on larvae and pupae of Bradysia impatiens. Conidia (ingestion-topical application) produced greater insecticidal action than insectistatic activity. Metabolites of B. bassiana had an insectistatic effect on the larvae; this mode of action is reported for the first time in this entomopathogenic fungus. Therefore, metabolites and conidia of B. bassiana have potential as biocontrol agents of B. impatiens populations.
