Introduction
Deforestation due to changes in land use and intensive use of some tree species modify the operation and structure of forest ecosystems, their recovery through natural processes is not always possible (Bustamante, Badano, & Pickett, 2012; Cayuela, Rey-Benayas, & Echeverría, 2006; Ramírez-Marcial, Camacho-Cruz, & González-Espinosa, 2008). At local level, changes are seen both under microclimatic and edaphic conditions and under the modification of other biotic interactions that affect seed germination and plant establishment (Bonfil & Soberón, 1999; Ramos-Palacios & Badano, 2014). Selective use of trees, at the highlands of Chiapas, has led to a reduction in the density and dominance of adult trees, mainly oaks (Quercus spp.), developing the regeneration and dominance of pines (Alba-López, González-Espinosa, Ramírez-Marcial, & Castillo-Santiago, 2003; Galindo-Jaimes, González-Espinosa, Quintana-Ascencio, & García-Barrios, 2002).
The restoration of degraded forest ecosystems is intended to restore their structure, function and productivity, as well as ecological processes and ecosystem services (Lamb, Stanturf, & Madsen, 2012; Stanturf, Palik, Williams, Dumroese, & Madsen, 2014; Torres-Miranda, Luna-Vega, & Oyama, 2011). Reforestation with multiple taxa is a viable option when woody species cannot be established naturally; this process allows to reconstruct the structure of the woody flora (Ramírez-Marcial et al., 2008).
Vegetation density, local abiotic environment conditions and existing disturbance regime are some factors that limit the establishment of species (Guo, Wang, Zhu, Wang, & Guo, 2011). For the purposes of restoration, it is necessary to understand the effect of climatic, edaphic and biotic conditions of the site on the survival and growth of the species used (Cardillo & Bernal, 2006; Pulsford, Lindenmayer, & Driscoll, 2016). Some Quercus species, due to their phenotypic plasticity, have potential to be used in forest restoration projects under different edaphic conditions (González-Espinosa et al., 2012) and canopy (Cardillo & Bernal, 2006; Ramírez-Marcial, Camacho-Cruz, González-Espinosa, & López-Barrera, 2006; Sánchez-Velásquez, Ramírez-Bamonde, Andrade-Torres, & Rodríguez-Torres, 2008). However, physical damage caused by grazing (Ramírez-Marcial, González-Espinosa, & García-Moya, 1996; Sánchez-Velásquez, Domínguez-Hernández, Pineda López, & Lara-González, 2011) water stress, degree of environmental disturbance, conditions of the substrate (Bonfil & Soberón, 1999; Flores-Cano, Badano, & Flores, 2012), and size and vigor of the seedlings (Bonfil, Rodríguez de la Vega, & Peña, 2000; Ramírez-Contreras & Rodríguez-Trejo, 2004) are factors that affect survival and growth.
An alternative that facilitates the establishment of the plants is the use of nurse shrubs or trees, since they improve microclimatic conditions and favor the initial growth (Bonfil & Soberón, 1999; Ramírez-Contreras & Rodríguez-Trejo, 2009; Ramírez-Marcial et al., 1996). Therefore, the yield analysis of plants along an environmental gradient, associated to human disturbance, allows to identify some barriers that prevent the establishment in sites with limited availability of propagules (Ramírez-Marcial et al., 2008; Ramos-Palacios & Badano, 2014).
The objective of this research was to evaluate the survival and growth of three oak species under three canopy conditions: forest, shrubland and grassland. The three species, characteristic of mountain ecosystems in southern Mexico and Guatemala, are in some category of endangered species (González-Espinosa, Meave, Lorea-Hernández, Ibarra-Manríquez, & Newton, 2011; Ramírez-Marcial et al., 2010). Quercus ocoteifolia Liebm., unlike Q. crispipilis Trel. and Q. segoviensis Liebm., is an evergreen species and requires canopy coverage conditions, cool temperatures and higher humidity for establishment (González-Espinosa et al., 2011; Gutiérrez & Trejo, 2014; Ramírez-Marcial et al., 2010). Therefore, the survival of Q. ocoteifolia is expected to be higher under forest canopy, while Q. crispipilis and Q. segoviensis have higher survival under more open canopy conditions.
Materials and methods
Study area
The study was carried out at the Parque Ecológico El Encuentro (PEE) located at the northeast portion of San Cristóbal de Las Casas (16° 43’ 54.72” - 16° 44’ 08.38” N and 92° 38’ 52.59” - 92° 38’ 24.52” W), at an average altitude of 2,270 m. The mean annual precipitation is 1,090.5 mm with an annual mean temperature of 15.0 °C (Comisión Nacional del Agua [CONAGUA], 2017). The PEE was under logging of Pinus for wood production and Quercus spp. and other broad-leaved species for firewood; it was also a sheep grazing site until 2010. The soil is moderately deep, derived from calcareous rocks and corresponding to Rendzina and Luvisol soils. The present vegetation includes secondary forests dominated by Pinus pseudostrobus Lindl., P. tecunumanii F. Schwerdtf. ex Eguiluz & J. P. Perry, Q. segoviensis, Q. crispipilis and Q. rugosa Née (De la Mora-Estrada, Ruiz-Montoya, Ramírez-Marcial, Morón-Ríos, & Mayorga-Martínez, 2017).
The study evaluated three canopy coverage conditions: pine-oak forest, Baccharis vaccinioides Kunth shrubland and grassland. A total of three plots of 100 to 150 m2 were established under each condition, which were considered experimental replications. The size varied according to the availability of the land, depending on the homogeneity of the coverage; grassland plots were the smallest and those of the forest were the largest. Conditions of canopy coverage, soil moisture and soil and air temperature were characterized in each plot. Canopy coverage was obtained from the analysis of six hemispherical photographs per plot, taken at 1 m height with a hemispherical lens (OptekaTM 0.20x, EUA) attached to a digital camera (Nikkon® modelo D5200, Singapur) and processed using the program HemiView (Rich, Wood, Vieglais, Burek, & Webb, 1999). Soil moisture and temperature were recorded by means of 20 readings per plot during the rainy season (October 2015) and 20 during the dry season (April 2016). Humidity was measured with Theta Meter® (model HH1, USA) and temperature with a digital dual-output thermometer J/K Extech Instruments® (model 421502, USA). Air temperature of each condition was continuously recorded one meter above the ground with a sensor HOBO TM® (Onset Computer Corporations, USA).
Plantation design
We used Q. crispipilis, Q. ocoteifolia and Q. segoviensis plants obtained from seeds collected in November and December 2013 and germinated in forest nurseries at El Colegio de la Frontera Sur (ECOSUR) in San Cristóbal de Las Casas. Quercus crispipilis and Q. segoviensis are found in Chiapas and Guatemala forming associations of pine-oak forests between 1,800 and 2,400 m, while Q. ocoteifolia is found in Oaxaca and Chiapas forming associations of pine-oak and cloud forest in wetter and cooler places, between 2,000 and 2,600 m (Ramírez-Marcial et al., 2010). Plants were kept for 15 months in the nursery and two months outside for their acclimatization. Root pruning in nursery was not applied. The three species were transplanted using a root ball during the last week of July 2015, to take advantage of the growth period (Ramírez-Marcial et al., 2006). A total of 33 plants per species were placed in each plot, randomly distributed at 1 m of equidistance. The amount per plot was determined based on the availability of plants of the three species studied.
Measurement variables
One week after transplantation, height and diameter of each plant were measured; these values were considered as the starting point of the experiment. Survival and growth assessments were repeated at 2, 4, 7, 9, 12, and 14 months after transplanting. Individual growth was measured through the maximum height and basal diameter of the stem. With these values, the relative growth rates (RGR) of both variables were calculated using the formula used by Hunt et al. (2002): RGR = [ln final growth (cm) - ln initial growth (cm)] / evaluation time (months).
At the end of the last evaluation (September 2016), biomass accumulated in stems, leaves and roots was quantified in a random sample of five plants per species and replication (a total of 135 plants). The collected plants were weighed per component; leaves, stem and root. Subsequently, the fractions were placed in an oven at 70 °C for 72 h for drying. At the end of this period, samples were weighed again using a scale with precision of 0.01 g, to obtain the dry weight.
Data analysis
A completely randomized design with 3 x 3 factorial arrangement (coverage and species type) was used. Compliance with the statistical assumptions of normality, homoscedasticity and independence was verified. Environmental variables among conditions were analyzed using the Kruskal-Wallis test; and comparison among pairs of coverage types, with the Wilcoxon rank test. The proportion of surviving plants was analyzed in each condition using the nonparametric Kaplan-Meier log rank test (Crawley, 2013). RGRs were analyzed, 14 months after transplantation, with ANOVA considering species and condition as main factors. Once significant differences among species (P < 0.05) were observed, another one-way ANOVA was used and the effect of the condition in each species was evaluated. Differences in biomass accumulated per fraction of each species, among conditions, were also evaluated with ANOVA. All analyzes were carried out using the program R version 3.2.2 (R Development Core Team, 2015).
Results and discussion
Microclimatic characteristics of sites
Table 1 shows the microclimatic conditions in the three coverage studied. The variables differed among transplant sites and seasons of the year; soil temperatures were significantly (P < 0.001) cooler in the forest. In autumn, soil moisture was similar (P = 0.32) in all three sites; however, at the beginning of spring, significant differences (P < 0.001) were observed with lower moisture in the shrubland compared to the forest and grassland.
Table 1.
| Microclimatic variable | Period | Coverage | Kruskal-Wallis X
|
|
||
|---|---|---|---|---|---|---|
| Forest | Shrubland | Grassland | ||||
| Soil temperature (°C) | October 2015 | 15.9 ± 0.05 a | 16.6 ± 0.12 b | 18.8 ± 0.08 c | 122.03 |
|
| April 2016 | 18.2 ± 0.18 a | 20.2 ± 0.20 b | 22 ± 0.24 c | 94.53 |
|
|
| Soil moisture (%) | October 2015 | 64 ± 1.59 a | 61 ± 1.83 a | 65 ± 1.63 a | 2.24 |
|
| April 2016 | 31 ± 1.43 a | 23 ± 1.03 b | 28 ± 1.47 a | 12.93 |
|
|
On the other hand, the mean air temperature was significantly lower (P < 0.05) in the forest and shrubland compared to the grassland (Figure 1). With regard to photosynthetically active radiation (PAR), the values were significantly (P = 0.05) higher for grassland, intermediate for shrubland and lower for forest (Figure 2).
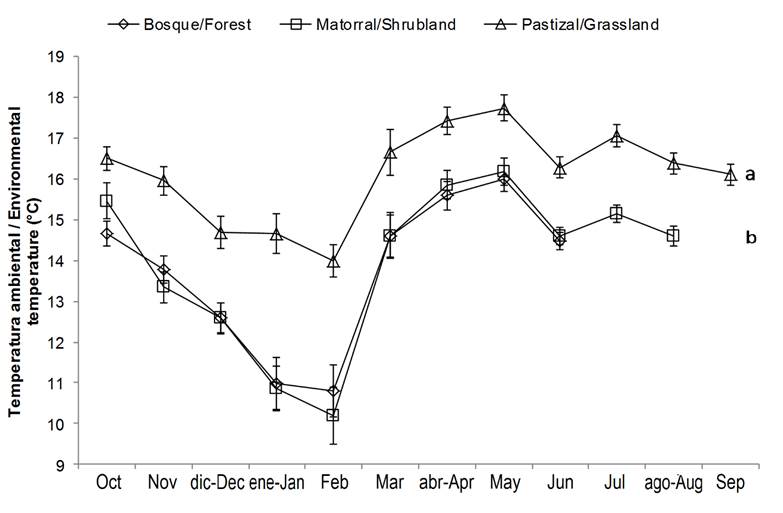
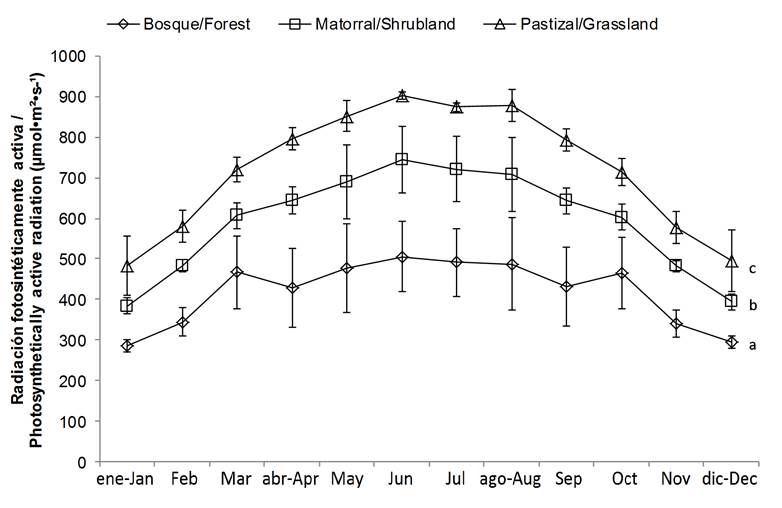
The results indicate that the microclimatic conditions vary according to the type of cover, which can affect the yield of the plants (Dickson, 1990). The absence of canopy is reflected in higher temperature, radiation and evapotranspiration, leading to considerable soil stress and desiccation, and lower plant growth (Arosa, Ceia, Costa, & Freitas, 2015; Ramírez-Marcial et al., 2008).
Survival of Quercus
Species survival after 14 months of evaluation was high under all three study conditions (> 90 %), except for Q. ocoteifolia (88 %) under grassland conditions (Figure 3). The follow-up period of the study allows to affirming that Quercus species had the ability to establish itself effectively under the three conditions; however, these trends can be modified based on the seasonality or growth stage of the plants used (Alvarez-Aquino & Williams-Linera, 2012; Espelta, Riba, & Retana, 1995).
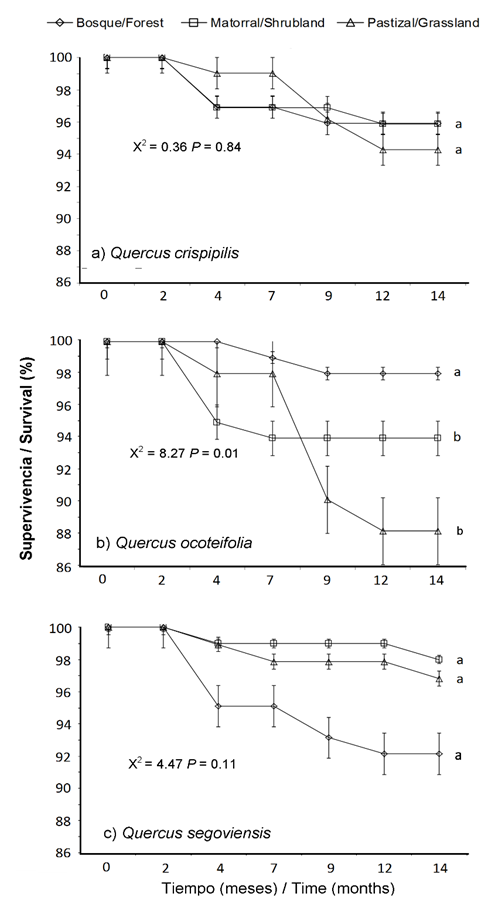
Short-term studies have shown that survival is influenced by microclimatic conditions created by the presence of trees, shrubs and native species of early succession. Such conditions may modify microbial and microclimatic conditions of the soil (Castro, Zamora, & Hódar, 2006; Ramírez-Contreras & Rodríguez-Trejo, 2009), improving the yield of plants under canopy conditions (Avendaño-Yáñez, Sánchez-Velázquez, Meave, & Pineda-López, 2014; Bonfil et al., 2000; Camacho-Cruz, González-Espinosa, Wolf, & de Jong, 2000; Castro et al., 2006).
Growth of Quercus
RGR and biomass differed significantly (P < 0.001) among species and canopy coverage (Table 2), suggesting a differentiated effect of the microenvironmental situations of each condition.
Table 2.
| Factor | Degrees of freedom | RGR height | RGR diameter | Biomass | |||
|---|---|---|---|---|---|---|---|
| Species | 2 | 15.52 | <0.001 | 13.82 | <0.001 | 19.22 | <0.001 |
| Site | 2 | 11.09 | <0.001 | 71.53 | < 0.001 | 17.85 | <0.001 |
| Species*Site | 4 | 1.59 | 0.17 | 4.45 | < 0.01 | 1.88 | 0.11 |
Figure 4 shows the RGR in height and diameter of the three Quercus species per coverage. RGR in height of Q. crispipilis and Q. ocoteifolia were higher in conditions with radiation and intermediate and low temperature (shrubland and forest). Quercus segoviensis had the lowest increase in RGR in height and showed no differences (P < 0.05) among sites. In the case of RGR in diameter, the three species increased at the site with the highest radiation (grassland) followed by shrubland and forest. This pattern was repeated in the accumulation of biomass in Q. crispipilis and Q. segoviensis, mainly in roots (Figure 5).
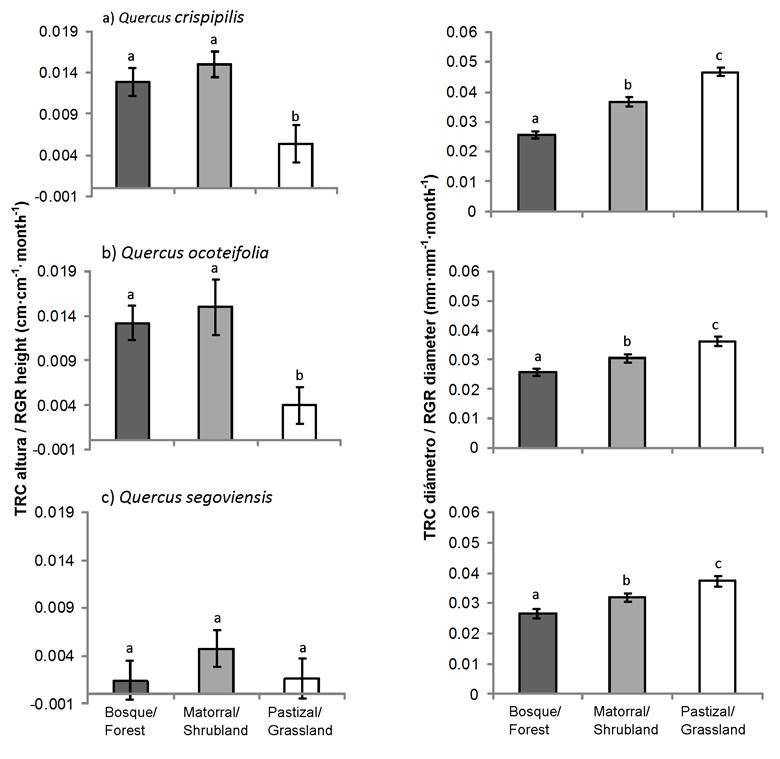
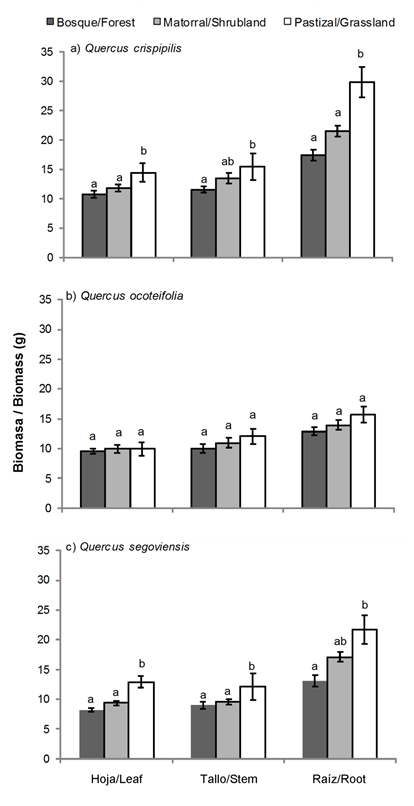
These results suggest a direct association between increased basal diameter and increased root biomass in Q. crispipilis and Q. segoviensis, in addition to an inverse relationship among RGR in height and basal diameter of Q. crispipilis and Q. ocoteifolia. The highest increases in height, in contrast to diameter, were associated with low and intermediate levels of solar radiation ranging from 5 to 50 % (Cardillo & Bernal 2006; Neufeld, 1983) and low temperature and humidity fluctuations under forest canopy (Padilla & Pugnaire, 2006; Ramírez-Contreras & Rodríguez-Trejo, 2009). Although light is considered a necessary resource for growth, increased radiation does not necessarily mean an increase in plant size (Espelta et al., 1995). The results indicate that, in oaks, the growth in basal diameter, associated with the increase in the radical biomass, is more common than the growth in height, shortly after transplanting, which coincides with that observed by Bonfil and Soberón (1999) and Kabeya and Sakai (2003).
The increase in biomass of each component (leaves, stem and root) of Q. crispipilis and Q. segoviensis was higher under grassland conditions compared to shrubland and forest conditions (Figure 5). The highest biomass increase in established plants at sites with higher solar radiation can be attributed to the fact that they suffer more stress, stem growth slows down and carbohydrates are distributed to lower parts (Dickson, 1990); in addition, under these conditions, plants reach higher photosynthetic rates (Feltrin et al., 2016; Kabeya & Sakay, 2003; Ramírez-Contreras & Rodríguez-Trejo, 2009). The component that reached the highest biomass was the root; the importance of this organ lies in the ability to store most of the carbohydrates the plant will use during periods of adverse conditions (Arosa et al., 2015, Kabeya & Sakay, 2003). The reserves stored in the roots allow oaks to regrow in case of death or partial removal of the aerial part (Bonfil & Soberón 1999; Cardillo & Bernal, 2006; Vázquez de Castro, Oliet, Puértolas, & Jacobs, 2014).
The three species of Quercus had high values of survival under the three experimental situations; although microclimatic variables are different among the studied conditions, it cannot be said that those are the only factors that affect survival, since herbivory and soil fertility can also influence them (Lei et al., 2013). As Q. crispipilis and Q. segoviensis showed higher biomass increases in the site with greater illumination, the use of these species in the restoration of abandoned grasslands is recommended; while Q. ocoteifolia would be more successful in restoring degraded secondary forests, because yield improves in the shade of forest canopy.
Conclusions
The presence of canopy influences the microclimatic variables of the sites; less coverage at the site will receive higher radiation and ambient and soil temperature. Despite the above, the survival of Q. crispipilis and Q. segoviensis was not affected by canopy coverage. As expected, relative growth rates of height and diameter changed according to the canopy coverage. The height of Q. crispipilis and Q. ocoteifolia was higher under forest and shrubland conditions; the diameter of the three species and the biomass of each component in Q. crispipilis and Q. segoviensis were higher under grassland, intermediate under shrubland and lower under the forest conditions. The three species seem to adapt to the different radiation conditions, temperature and humidity of each site. Quercus crispipilis and Q. segoviensis have better survival and growth responses under shrubland and grassland conditions, while Q. ocoteifolia is favored by the presence of forest canopy.
